Autoimmune Aspects of Neurodegenerative Diseases
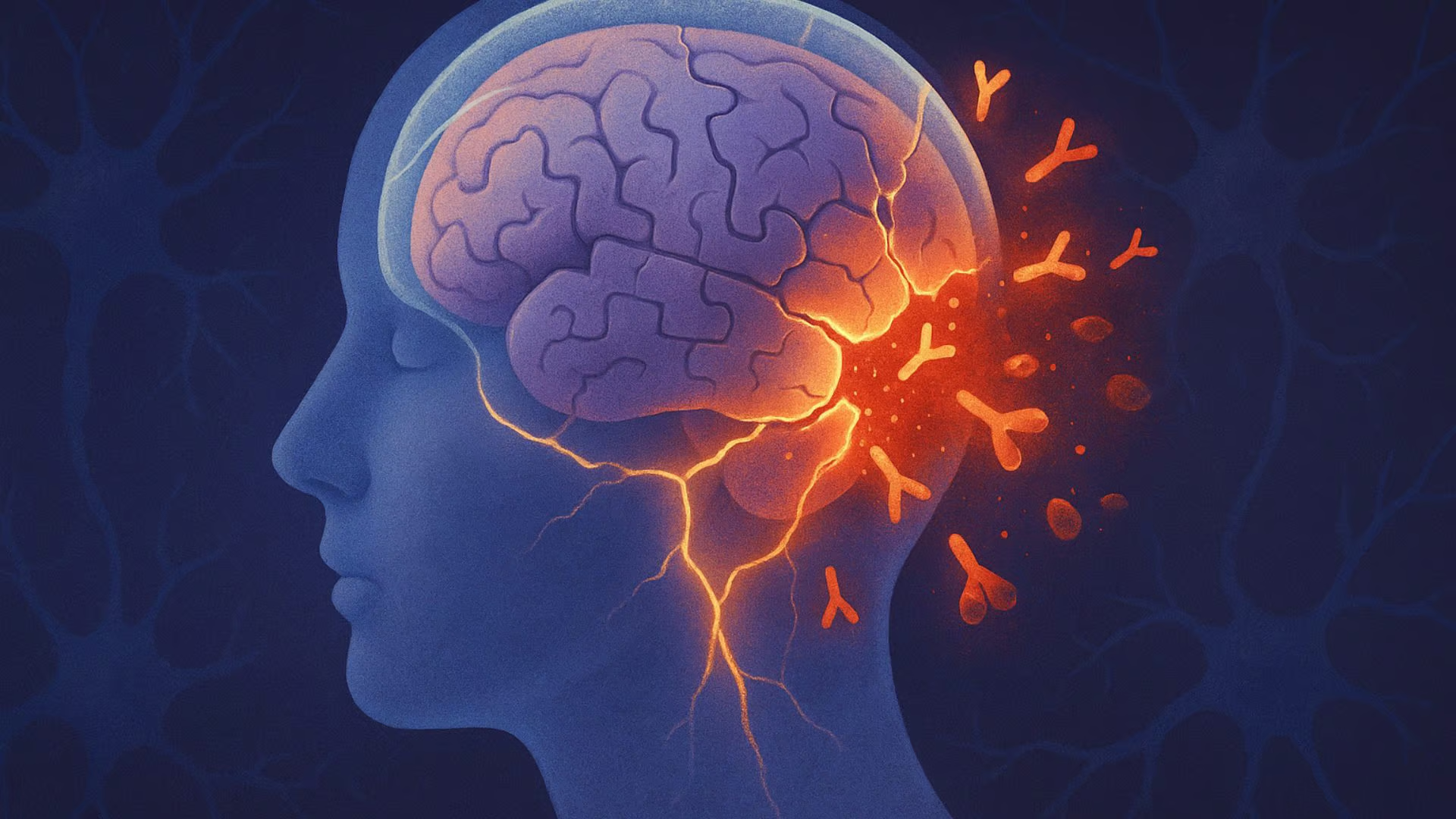
Alzheimer’s disease and Parkinson’s disease have generally been thought of as neurodegenerative diseases, meaning those that damage and destroy parts of the brain. However, evidence now suggests that immune system dysfunction plays a role in these conditions, giving them autoimmune characteristics as well. Multiple sclerosis (MS), already established as a neurodegenerative autoimmune disease, offers a contrast to the emerging immune-related findings in Parkinson’s and Alzheimer’s disease.
Experts in the field of neurodegeneration and neuroinflammation, Alessandro Sette, a professor and director of the Center for Autoimmunity and Inflammation at La Jolla Institute for Immunology, Esther Melamed, M.D., Ph.D., a neurologist and assistant professor at the Dell Medical School at The University of Texas at Austin, and Dr. Donald Weaver of the Krembil Research Institute discuss how the recognition of these diseases as autoimmune can change how they’re diagnosed and treated in the future.
What Evidence Links Neurodegenerative Disease to Autoimmunity?
Multiple Sclerosis: The Established Neurodegenerative Autoimmune Disease
MS involves an autoimmune attack on myelin, the insulating sheath surrounding nerve cells in the brain and spinal cord. When myelin is compromised, it is more difficult for neurons to communicate with one another, which can lead to nearly any neurological symptom.
In 2024, a research study involving over 10 million individuals found that nearly 10% of MS cases have distinct sets of autoantibodies years before symptoms appear (1). These autoantibodies target a shared molecular pattern found in both human and viral proteins, including those from Epstein-Barr Virus, a virus linked to MS in 2022 (2).
It’s possible that in the future, a blood test could help identify multiple sclerosis risk before any symptoms appear.
“The hope is when people start getting treated early on for their neuroinflammatory MS condition that it would slow down neurodegeneration and hopefully prevent it,” says Melamed.
Parkinson’s Disease: When T Cells Attack Neurons
In Parkinson’s disease, brain cells that produce dopamine die off, giving rise to motor and non-motor symptoms, including tremors, rigidity, and balance issues. The symptoms in Parkinson’s develop gradually. “Initially, we don’t really have the symptoms that we all associate with Parkinson’s, such as cognitive and motor symptoms,” says Sette.
“There’s a process of destruction that goes unnoticed.”
Sette first found evidence of the autoimmune aspects of this disease in 2017 when he and collaborators at Columbia University discovered that T cells recognize alpha-synuclein peptides, which aggregate in Parkinson’s disease, and initiate an autoimmune attack on neurons (3). The following year, work from another group found that T cells from people with Parkinson’s disease attack dopamine-producing neurons (4). On the other hand, T cells from people without Parkinson’s did not.
While Sette has found that T cells target alpha-synuclein in Parkinson’s disease, not all patients have T cells against it. Therefore, his team searched for other T cell targets in the disease. In 2024, they found another target: a protein called PINK1 (5). PINK1 helps the brain regulate mitochondria, which provide energy for the cell. When T cells attack cells that express PINK1, it leads to inflammation and neuronal death. Despite these molecular clues, Sette says it’s not clear whether autoimmune reactivity is the culprit behind the disease or if there’s another cause of damage responsible for the initial inflammation.
Alzheimer’s Disease: Is It an Innate Autoimmune Disorder?
Research into the autoimmune aspects of Alzheimer’s disease is still limited compared to multiple sclerosis and Parkinson’s disease, but some key insights are beginning to emerge.
One of the main features of Alzheimer’s is the buildup of amyloid beta, a protein that, in high numbers, clumps together and forms plaques around brain cells.
While the normal function of amyloid beta is unclear, it appears to play a role in protecting the brain from invading microbes by triggering an innate immune response, the body’s first line of defense. According to Dr. Weaver, where problems arise is when amyloid beta’s antimicrobial properties result in a “misdirected attack upon ‘self’ neurons” (6). Weaver suggests the misdirected attack may result from electrophysiological similarities between neurons and bacteria.
Blood-Based Biomarkers May Predict Autoimmune Risk
In 2018, scientists realized that patients with ulcerative colitis and other autoimmune disorders had a lower risk of developing Parkinson’s disease (7). A commonality that the researchers found among these patients were the immunosuppressive drugs used to treat these diseases and the researchers suggest that these drugs could lower risk of Parkinson’s disease.
But does it make sense to treat people preemptively with immunosuppressants – which can be expensive and have side effects – to avoid developing Parkinson’s disease? That depends on the patient’s risk. Sette explains, “if you could have a test that says, ‘aha, you are at high risk of being on a pathway of progression towards Parkinson,’ then I think it would be justified to intervene to shut down this festering inflammation.”
A person’s blood could give clues into future autoimmune diseases. For multiple sclerosis, signs of disease could be found in the blood years before MS onset (8). The same can be seen in Parkinson’s disease. Sette and colleagues found that alpha-synuclein reactive T cells could be found in the blood of Parkinson’s patients years before a Parkinson’s diagnosis (9).
“It becomes a question of if we can develop biomarkers of neuroinflammation which potentially may contribute to or worsen neurodegeneration and if we can come up with the right people to screen from early on,” says Melamed. “Perhaps they have family history or perhaps they have other autoimmune conditions. We might be able to slow down or stop this process that’s starting before it shows up.”
Genetic Connections Between Autoimmunity and Neurodegeneration
HLA, or human leukocyte antigen, is a molecule that’s expressed on the surface of certain immune cells that help the immune system determine self from non-self. Mutations in HLA genes have been connected with various autoimmune diseases.
“HLA certainly is the biggest risk factor for autoimmune conditions and neurodegenerative conditions and some of that is thought to be driven by microglial dysfunction,” says Melamed.
Microglia help clear damaged proteins or aggregated proteins in the brain and the HLA molecule on these cells help them recognize what to target. Aside from HLA, Melamed notes that hundreds of other genes have been identified that affect both autoimmunity and neurodegeneration such as those involved in blood brain barrier integrity, mitochondrial genes, and immune regulatory genes. One study from 2017 found 17 genes in common between Parkinson’s and other autoimmune diseases including multiple sclerosis, Crohn’s disease, and rheumatoid arthritis, among others (10).
In the last decades, advances in genomic and proteomic technologies have allowed scientists to more quickly and more cheaply identify trends in the genes and protein associated with disease among a large patient population.
This allows scientists to look at and analyze multiple biomarkers of disease at once.
Genomics and proteomics are most used in the research setting but it’s possible that in the future, these technologies could be put into play in personalized medicine, where a person’s genetic profile could be used to make medical decisions.
Managing Modifiable Risk Factors for Brain and Immune Health
While a person’s genes are inherited, many other external lifestyle factors impact the progression of autoimmune diseases. Melamed, who studies how genetics, diet, and gut microbiome influences multiple sclerosis says, “[lifestyle] is the one factor that it’s in our control.” She notes that vitamin deficiency and poor sleep has been linked to a higher risk of both autoimmune diseases and neurodegenerative diseases.
Melamed says, “the best thing we can do for ourselves… is to make sure that we get enough sleep, avoid smoking and heavy alcohol use, manage stress, prioritize exercise, and keep a healthy diet.”

About the Author
Sources
- Article Sources
Zamecnik, C. R., et al. (2024). An autoantibody signature predictive for multiple sclerosis. Nature medicine, 30(5), 1300–1308. https://doi.org/10.1038/s41591-024-02938-3
Bjornevik, K., et al. (2022). Longitudinal analysis reveals high prevalence of Epstein-Barr virus associated with multiple sclerosis. Science (New York, N.Y.), 375(6578), 296–301. https://doi.org/10.1126/science.abj8222
Sulzer, D., et al. (2017). T cells from patients with Parkinson’s disease recognize α-synuclein peptides. Nature, 546(7660), 656–661. https://doi.org/10.1038/nature22815
Sommer, A., et al. (2019). Th17 Lymphocytes Induce Neuronal Cell Death in a Human iPSC-Based Model of Parkinson’s Disease. Cell stem cell, 24(6), 1006. https://doi.org/10.1016/j.stem.2019.04.019
Williams, G. P., et al. (2024). PINK1 is a target of T cell responses in Parkinson’s disease. The Journal of clinical investigation, 135(4), e180478. https://doi.org/10.1172/JCI180478
Weaver D. F. (2023). Alzheimer’s disease as an innate autoimmune disease (AD2): A new molecular paradigm. Alzheimer’s & dementia : the journal of the Alzheimer’s Association, 19(3), 1086–1098. https://doi.org/10.1002/alz.12789
Racette, B. A.,et al. (2018). Immunosuppressants and risk of Parkinson disease. Annals of clinical and translational neurology, 5(7), 870–875. https://doi.org/10.1002/acn3.580
Zamecnik, C. R., et al. (2024). An autoantibody signature predictive for multiple sclerosis. Nature medicine, 30(5), 1300–1308. https://doi.org/10.1038/s41591-024-02938-3
Lindestam Arlehamn, C. S., et al. (2020). α-Synuclein-specific T cell reactivity is associated with preclinical and early Parkinson’s disease. Nature communications, 11(1), 1875. https://doi.org/10.1038/s41467-020-15626-w
Witoelar, A., et al. Genome-wide Pleiotropy Between Parkinson Disease and Autoimmune Diseases. JAMA neurology, 74(7), 780–792. https://doi.org/10.1001/jamaneurol.2017.0469